A potential cure for HIV just got approval from the FDA to begin human trials.
American Gene Technologies (AGT), a biotechnology company in Maryland, said that it got approval to move forward with trials for a gene therapy protocol that it believes will eliminate the virus in people living with HIV.
Related: Anti-LGBTQ Christian hate group cheers as Trump administration shuts down HIV cure research
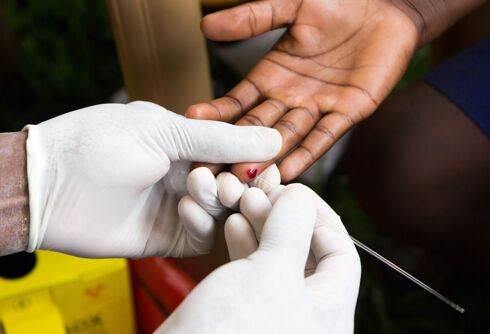
The trial is for AGT103-T, a cell product that results from an 11-day process that involves extracting blood from a person living with HIV and separating the T cells, which are involved in the body’s immune response, according to the Washington Blade.
Never Miss a Beat
Subscribe to our newsletter to stay ahead of the latest LGBTQ+ political news and insights.
The T cells are then genetically altered in a lab to make them immune to HIV so that they can fight the virus. The cells also multiply in the lab and are then put back in the person they were taken out of. AGT hopes that they will then clear the body of the virus.
“This is momentous news that we have FDA approval to launch Phase 1 and conduct our first human trials,” said AGT’s chief science officer David Pauza in a statement.
“We are beyond excited to reach this milestone. This brings us closer to our goal of transforming lives with genetic medicines.”
“I am confident AGT103-T will be an important step towards an eventual cure for HIV,” said AGT CEO Jeff Galvin.
He said that the cost of the treatment is around $200,000, but he believes that the price will go down as the procedure becomes more common and an automated process is developed.
Phase 1 of the trial will test AGT103-T for safety and will take place in the D.C. area.
Don't forget to share: